| Enterprise Solutions | Instrument Services | Parts & Consumables | Support | Training | About Us | Contact Us |
USA phone for analytical instruments: 800-532-4752
USA phone for laboratory equipment: 1 888-860-5227, Option 3, then 1
Need contact info for other countries? Click here.

Leveraging Fishbone Diagrams to Uncover Root Cause
By Kelly Huckabone
April 2020
The Fishbone Diagram or Ishikawa Diagram is a cause-and-effect diagram that helps project teams identify root causes for issues that can include but are not limited to imperfections, variations, defects, and/or failures.
The diagram looks just like a fish's skeleton with the problem at its head and the causes for the problem feeding into the spine. There are different iterations of the fishbone (different spines of the fish).
At Thermo Fisher Scientific, we use the 4 Ms and E Approach. We brainstorm inputs from material, methods, manpower, machinery and environment and once the exercise is completed, we look at the spine of the fish that has the most concentration of inputs and that helps identify areas to focus on for root cause analysis. It validates the area of the problem and helps identify where the team needs to pull data from to help further define the root cause. Often, there’s a symbiotic relationship between the identified root cause and the containment and/or corrective action measures.
Below is a great example from a recent project. The team brainstormed all of the possible root causes and there was a heavier concentration on the “Methods” spine. The team assessed the root cause and then looked at solutions around addressing the key issues identified under the “Methods” spine.
The Fishbone Diagram exercise requires a strong facilitator to help employees feel comfortable in expressing their ideas. Sometimes, some of the cause identified fit under more than one spine. We try to find the best spine to reflect the issue. As part of our continual improvement project, most formal projects require our teams to complete some formal method for root cause analysis. The Fishbone Diagram is one that works well in a cross-functional and complex project.
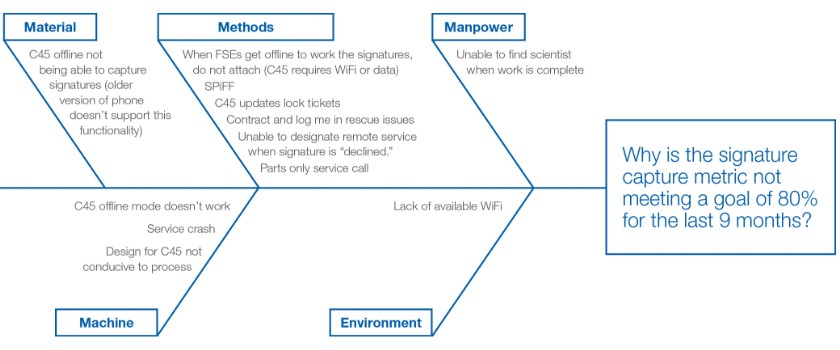
Root cause: Difficult to capture signatures due to platform issues and customer availability (focus on Method)
Download a PDF of this article